 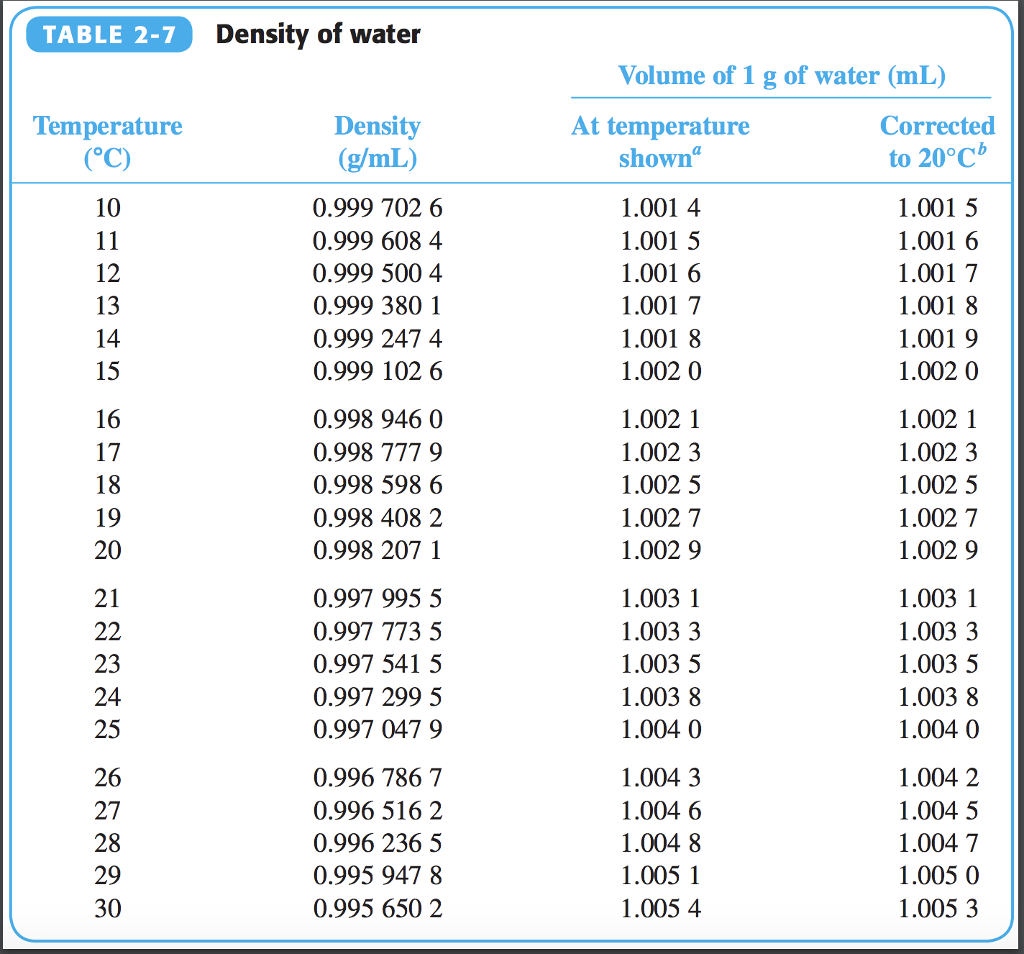 
That's why the difference between heat of vaporization at 25C (energy required to break all H-bonds between 1 gram of initially slow moving molecules) and at 100C (energy to break all H-bonds of 1 gram of fast molecules) is LESS then the energy required to make all of those 1 gram of molecules faster. Explore the benefits of water density for living creatures and the examples of seawater density. And as more molecules fly off, less energy is needed to break off the remaining bonds. Learn how the density of water varies with temperature, purity and depth, and why ice floats on water. The specific volume of water depends on temperature as shown below: Heavy. The density of distilled water is the same. At room temperature, water remains in a liquid state. The density of water at 25 degrees Celsius is 997 kg/m3. At room temperature ( 200C), its value is 998.2 kg/m3. The density of water is defined similarly to other substances. Temperatur must be within the ranges 0-370 ☌, 32-700 ☏, 273-645 K and 492-1160 °R. The Density of Water At Room Temperature. The output specific volume is given as cm /g, ft /lb, gal (US liq)/lb and ft. It occurs more and more as you near towards it. The calculator below can be used to calculate the liquid water specific volume at given temperatures. You don't need to wait until 100 degrees for vaporization to begin. For example, the density of ultrapure water at 20.00 ☌ is known to be 0.998203 g/cm 3: any deviation from this value ± tolerances would imply that the water sample contains impurities. Remember that when you apply energy to water, some of it will increase the avg kinetic energy of the molecules (related to the temperature) and some will be spent to break off all hydrogen bonding and send the molecules flying away (related to heat of vaporization at a GIVEN temperature). The measurement of density is crucial in different industries to ensure quality for both raw materials and finished goods. Try finding a household item more dense than water it is more difficult than you may think Practice Problems. How come only 40 calories can increase the water temperature by 75 degrees to its boiling point, if the specific heat property tells us that 40 calories can only increase it by 40 degrees? An item will only sink in water if it is more dense than water. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
