 The reason for this is that hydrogen, under normal pressures and temperatures, actually behaves like a nonmetal. There is one exception to this, which is Hydrogen (symbol: H, number 1). Then, you will find the non-metals (those elements where electrons outnumbered protons) in the right area.If you look at the periodic table, you will find all the elements with a positive ionic charge (those where protons outnumbered electrons) in the left area.Electrons are particles with a negative charge, while protons are particles with a positive charge. Ionic charges, which is sometimes merely known as “charges” are the difference between the number of electrons and the number of protons. Instead, the elements are arranged by their atomic numbers. The main difference between the current periodic table has one major difference with the Mendeleev periodic table is that now the elements are no longer organized by their mass number. These symbols and numbers work as shortcuts to identify them. Each of these elements has a symbol and an atomic number. The current version of the periodic table contains 118 different chemical elements. Pt, Ir, Os) or which increase regularly (eg. In his version of the periodic table (known as the Mendeleev periodic table), the elements are organized by mass number, in increasing order.Įlements which are similar as regards their chemical properties have atomic weights which are either of nearly the same value (eg. That first version contained 33 elements, which Lavoisier had grouped into the following categories:Īfter undergoing many changes through the 19 th century, it was not until 1869 than a Russian chemist called Dmitri Mendeleev came up with his own table in 1869. The first known attempt at organizing all chemical elements into a tabular arrangement dates back to 1789 when French chemist Antoine Lavoisier. The modern periodic table is the result of hundreds of years’ worth of research. We are made of star stuff.” - Carl Sagan What Is The Periodic Table? 
“The nitrogen in our DNA, the calcium in our teeth, the iron in our blood, the carbon in our apple pies, we are made in the interior of collapsing stars. In other words, we start off by learning the periodic table but then we must also learn all the other relevant details about each and every one of the elements. The next step would be to add the ionic charges, the full names, and the mass of each of the elements. It is a merely first step before going deeper into it. But just learning the periodic table is not really enough. 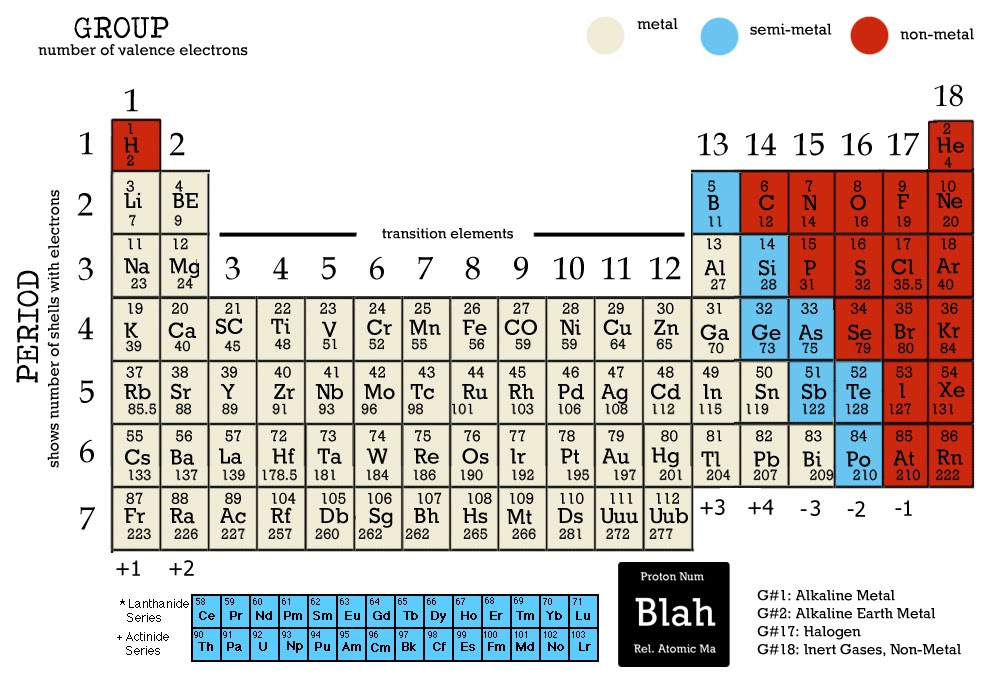
There can be no doubt that any science student who is interested in chemistry must learn the periodic table. The periodic table can also be broken down by name and mass depending on your interests. The periodic table ionic charge can be broken down by metals that are positive and on the left of the table and nonmetals which are negative and found on the right. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
